JamiiForums
JF Official Account
- Nov 9, 2006
- 6,202
- 5,012
MARADHI YA KIHARUSI
Kiharusi ni hali inayotokea pale mishipa inayosambaza damu kwenye ubongo inapopatwa na misukosuko kiasi cha kushindwa kupeleka damu kwenye ubongo na hivyo kuathiri ufanyaji kazi wa ubongo kwa zaidi ya masaa 24.
Kulingana na shirika la afya duniani kiharusi ni nakisi katika neva ambayo hutokana na tatizo katika mishipa ya damu ya ubongo na ambalo huendelea zaidi ya masaa 24 au kukoma ndani ya masaa 24. Muda wa masaa 24 umechukuliwa ili kutofautisha kiharusi na kiharusi cha kukosa oksijeni katika ubongo (ischemia) ambacho hutokea na kudumu kwa muda mfupi yaani (transient ischemic attack).
Kwa kawaida dalili za TIA hupotea na mtu kurudia hali yake ya kawaida ndani ya masaa 24.

Aina za kiharusi
Kiharusi kimegawanyika katika aina kuu mbili kulingana na jinsi kinavyotokea (visababishi vyake). Aina hizo ni:
1. Kiharusi cha kukosa hewa kwenye Ubongo (Ischemic stroke)
Aina hii ya kiharusi hutokea iwapo usambazaji wa damu katika sehemu ya ubongo hupungua na kupelekea tishu za ubongo za eneo liliathirika kushindwa kufanya kazi zake vizuri.
Aina hii ya kiharusi husababishwa na nini?
Aina hii ya kiharusi husababishwa na damu iliyoganda kuziba mishipa midogo ndani ya ubongo (cerebral thrombosis) au Kipande cha damu kumeguka na kwenda kuziba mshipa wa damu kwenye ubongo(cerebral embolism)
Kupungua kwa usambazaji wa damu kwa ujumla kwa mfano shock.
Vena thrombosis.
2. Kiharusi cha kuvuja damu ndani ya Ubongo (Haemorrhagic Stroke)
Aina hii ya kiharusi hutokea pale ambapo mishipa midogo ya damu ndani ya ubongo hupasuka na kusababisha kusambaa kwa damu ubongoni (cerebral hemorrhage). Mara nyingi aina hii ya kiharusi hutanguliwa na dalili za kichwa kuuma, au kuwepo kwa historia ya ajali ya kichwa.
Visababishi vya Kiharusi (kwa ujumla)
- Wakati fulani, mishipa ya damu huzungukwa na aina fulani za mafuta yasiyofaa mwili, hali inayoitwa kitaalamu kama atherosclerotic plaque. Hali hii husababisha kuganda kwa damu katika
- mishipa (thrombosis) hiyo. Kuganda huku kwa damu kunaweza kutokea kwenye mishipa mikubwa au midogo inayopeleka damu kwenye ubongo. Baadhi ya mishipa mikubwa ya damu inayoweza
- kuathirika na tatizo hili ni pamoja na arteri za common carotid na interior carotid arteries, na arteri za vertebral. Nyingine ni mishipa midogo inayounda eneo linaloitwa circle of willis lililo katika ubongo.
- Kuganda kwa chembe nyekundu za damu za mgojwa wa sickle cell kunaweza pia kusababisha kuziba mishipa ya damu hivyo kusababisha Kiharusi.
- Kuganda kwa damu (embolus) kwenye mishipa ya damu ya sehemu nyingine za mwili au (hewa, mafuta, kusanyiko la vijimelea kama bakteria waletao ugonjwa wa endocarditis) huweza
- kusababisha kuziba kwa mishipa inayosambaza damu kwenye ubongo na hivyo kusababisha kiharusi.
- Upungufu wa usafirishaji damu mwilini (systemic hypoperfusion) - Wakati fulani moyo hushindwa kusukuma damu vizuri kwenda sehemu mbalimbali za mwili ikiwemo ubongo kutokana na kufa kwa
- sehemu za nyama ya moyo kwa kukosa damu ya kutosha (Ischaemic Heart Diseases) au kutokana na kujaa kwa maji kwenye gamba lake la nje (pericardial effusion) au kupungua kwa damu mwilini,
- kunakoweza kusababishwa na mambo kadhaa. Hali hii husababisha sehemu kubwa ya mwili ikiwemo ubongo kukosa oksijeni ya kutosha na kusababisha kuathiriwa kwa sehemu ya ubongo.
- Shinikizo la damu la muda mrefu lisilothibitiwa linaweza kusababisha kupasuka kwa mishipa midogo inayosambaza damu kwenye ubongo na hivyo damu kujaa kwenye ubongo (Intracerebral hemorrhage).
Dalili za kiharusi
Dalili za kiharusi hutegemea na eneo la ubongo liliathiriwa.
- Iwapo sehemu ya ubongo mkubwa ( Cerebellum) itaadhirika, mgonjwa atakuwa na dalili kama kushindwa kutembea vizuri, hivyo kuathiri mwendo wake, kujihisi kizunguzungu na pia kutapika.
- Iwapo sehemu ya ubongo wa kati (cerebral cortex) itakuwa imeathirika, mgonjwa atakuwa na dalili za kushindwa kuongea,kushindwa kuelewa lugha inayozungumzwa, kushindwa kuona vizuri, kuwa
- na ukosefu wa kumbukumbu, kuwa na mvurugiko wa mpangilio wakw wa kufikiri, kuchanganyikiwa, na kubadilika kwa mwendo wa harakati za hiari.
- Iwapo mgonjwa ataadhirika sehemu ya ubongo kwenye shingo kuelekea kwenye uti wa mgongo (brain stem), mgonjwa anaweza kuwa na dalili za kuhisi mabadiliko ya harufu, ladha, kusikia na
- kuona; kulegea kwa misuli ya macho (ptosis); kupungua kwa ufahamu na kulegea kwa misuli ya uso; Ulegevu wa Ulimi (kushindwa kutoa nje au kusogeza upande upande); kupungua uwezo wa
- kumeza; ulegevu wa misuli ya shingo na kushindwa kugeuza shingo upande mmoja; kushindwa kusimama sawasawa na kuona vitu kwa hali ya utofauti; mabadiliko ya upumuaji na kiwango cha moyo kudunda.
- Iwapo sehemu mojawapo ya mfumo mkuu wa neva (central nervous system) imeathirika, mgonjwa atakuwa na dalili za kupoteza ufahamu kwa upande mmoja wa mwili na kulegea kwa misuli ya uso, kuhisi ganzi mwilini, na kupungua kwa ufahamu wa hisia na hisia mtetemo.
- Aidha bila kujalisha eneo lililoathiriwa, mgonjwa pia anaweza kuwa na dalili za kupoteza fahamu, maumivu makali ya kichwa na kutapika. Dalili hizi za kuumwa kichwa na kutapika kwa kawaida
- hutokea kwa mgonjwa mwenye kiharusi cha kuvuja damu (hemorrhagic stroke) ambacho husababisha ongezeko la shinikizo na mgandamizo wa ubongo ndani ya fuvu kutokana na kuvuja kwa damu.
Vipimo
ECG, ECHOCARDIOGRAM huwezesha kutambua hitilafu katika mapigo ya moyo (arrhythmia) na kama kuna damu iliyoganda kwenye moyo ambayo inaweza kufika kwenye ubongo
Holter monitor husaidia kutambua hitilafu katika mapigo ya moyo (arrhythmia) zinazotokea kwa vipindi
Angiogram huwezesha kugundua matatizo kwenye mishipa ya damu, na ni mishipa ipi ya damu iliyoziba.
Vipimo vya damu huwezesha kutambua uwepo wa lijamu mwilini (hypercholesterolemia) na mabadiliko mengine katika damu
Vihatarishi vya kiharusi
Vitu vinavyoweza kumuweka mtu katika hatari ya kupata kiharusi ni pamoja na
Matibabu ya kiharusi hutegemea pia aina na visababishi vyake .
1. Matibabu ya kiharusi kinachotokana na ubongo kukosa hewa (Ischemic stroke)
Mgonjwa hutibiwa kwa kutumia dawa za kuyeyusha damu iliyoganda (thrombolytics) au kwa kuondoa damu iliyoganda kwa njia mbalimbali (thrombectomy). Dawa nyingine kama vile junior Aspirin na Clopidogrel hutolewa kwa ajili ya kuzuia chembe sahani kukusanyika na kuganda.
2. Matibabu ya kiharusi kinachotokana na damu kuvujia kwenye ubongo (Hemorrhagic stroke)
Aina hii ya kiharusi huitaji tathmini ya upasuaji wa neva ili kuchunguza na kutibu sababu ya damu kuvuja. Angalizo: ni hatari kumpa mgonjwa wa aina hii ya kiharusi dawa za kuyeyusha damu
iliyoganda au za kuzuia kuganda maana uhatarisha maisha ya mgonjwa badal ya kumsaidia. Kwahiyo ni vizuri kwa wataalamu kufanya ufanya uchunguzi wa kutosha ili kuwa na uhakika na tatizo.
Huduma na Matunzo kwa mgonjwa wa Kiharusi
Mojawapo ya mambo muhimu ya kufanya kwa mgonjwa aliyepata kiharusi ni kumuongoza na kumuelimisha ili arudishe ujuzi wake wa maisha ya kila siku. Hapa uhitaji ushirikiano wa wauguzi,
wataalamu wa viungo, wataalamu wa ushauri wa kazi na daktari. Kuna umuhimu pia wa kuwaelimisha ndugu kuhusu hali ya mgonjwa ili waweze kumsaidia katika matunzo yake nyumbani na kwenye jamii inayomzunguka.
Matarajio (prognosis)
Asilimia 75 ya wagonjwa wa kiharusi wanaonusurika kifo huwa walemavu na kusababisha kuathirika kwa ufanyaji kazi wao na kuajiriwa. Ulemavu unaweza kuwa wa kimwili au kiakili au vyote kwa pamoja.
Ulemavu wa kimwili ni pamoja na
- Ulegevu wa misuli
- Kujihisi ganzi sehemu mbalimbali za mwili hususani zilizoathirika
- Kujikojolea
- Kutoona vizuri
- Kushindwa kuendelea na shughuli za kila siku
- Vichomi
- Vidonda shinikizo
- Kukosa hamu ya chakula.
- Ulemavu wa akili hujumuisha vitu kama
- Mgonjwa kuwa na wasiwasi
- Hofu ya mashambulizi, na
- Unyongovu.
Jinsi ya kuzuia kiharusi
- Zuia au tibu shinikizo la damu
- Dhibiti kisukari
- Fanya mazoezi
- Acha kuvuta sigara
- Punguza uzito
- Acha kunywa pombe kupita kiasi
- Tumia Junior Aspirin
- Kula chakula kisichokuwa na mengi na kisicho na chumvi nyingi.
- Tumia dawa za kupunguza mafuta mwilini (statins)kwa mfano Simvastatin
===
What Is a Stroke?
Stroke is a medical emergency and a leading cause of death in the U.S. It occurs when a blood vessel in the brain bursts or, more commonly, when a blockage develops. Without treatment, cells in the brain quickly begin to die. The result can be serious disability or death. If a loved one is having stroke symptoms, seek emergency medical attention without delay.

Stroke Symptoms
Signs of a stroke may include:

Stroke: Time = Brain Damage
Every second counts when seeking treatment for a stroke. When deprived of oxygen, brain cells begin dying within minutes. There are clot-busting drugs that can curb brain damage, but they have
to be used within three hours of the initial stroke symptoms. Once brain tissue has died, the body parts controlled by that area won't work properly. This is why stroke is a top cause of long-term disability.
Kiharusi ni hali inayotokea pale mishipa inayosambaza damu kwenye ubongo inapopatwa na misukosuko kiasi cha kushindwa kupeleka damu kwenye ubongo na hivyo kuathiri ufanyaji kazi wa ubongo kwa zaidi ya masaa 24.
Kulingana na shirika la afya duniani kiharusi ni nakisi katika neva ambayo hutokana na tatizo katika mishipa ya damu ya ubongo na ambalo huendelea zaidi ya masaa 24 au kukoma ndani ya masaa 24. Muda wa masaa 24 umechukuliwa ili kutofautisha kiharusi na kiharusi cha kukosa oksijeni katika ubongo (ischemia) ambacho hutokea na kudumu kwa muda mfupi yaani (transient ischemic attack).
Kwa kawaida dalili za TIA hupotea na mtu kurudia hali yake ya kawaida ndani ya masaa 24.
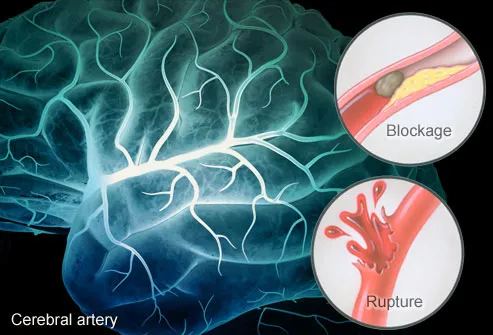
Aina za kiharusi
Kiharusi kimegawanyika katika aina kuu mbili kulingana na jinsi kinavyotokea (visababishi vyake). Aina hizo ni:
1. Kiharusi cha kukosa hewa kwenye Ubongo (Ischemic stroke)
Aina hii ya kiharusi hutokea iwapo usambazaji wa damu katika sehemu ya ubongo hupungua na kupelekea tishu za ubongo za eneo liliathirika kushindwa kufanya kazi zake vizuri.
Aina hii ya kiharusi husababishwa na nini?
Aina hii ya kiharusi husababishwa na damu iliyoganda kuziba mishipa midogo ndani ya ubongo (cerebral thrombosis) au Kipande cha damu kumeguka na kwenda kuziba mshipa wa damu kwenye ubongo(cerebral embolism)
Kupungua kwa usambazaji wa damu kwa ujumla kwa mfano shock.
Vena thrombosis.
2. Kiharusi cha kuvuja damu ndani ya Ubongo (Haemorrhagic Stroke)
Aina hii ya kiharusi hutokea pale ambapo mishipa midogo ya damu ndani ya ubongo hupasuka na kusababisha kusambaa kwa damu ubongoni (cerebral hemorrhage). Mara nyingi aina hii ya kiharusi hutanguliwa na dalili za kichwa kuuma, au kuwepo kwa historia ya ajali ya kichwa.
Visababishi vya Kiharusi (kwa ujumla)
- Wakati fulani, mishipa ya damu huzungukwa na aina fulani za mafuta yasiyofaa mwili, hali inayoitwa kitaalamu kama atherosclerotic plaque. Hali hii husababisha kuganda kwa damu katika
- mishipa (thrombosis) hiyo. Kuganda huku kwa damu kunaweza kutokea kwenye mishipa mikubwa au midogo inayopeleka damu kwenye ubongo. Baadhi ya mishipa mikubwa ya damu inayoweza
- kuathirika na tatizo hili ni pamoja na arteri za common carotid na interior carotid arteries, na arteri za vertebral. Nyingine ni mishipa midogo inayounda eneo linaloitwa circle of willis lililo katika ubongo.
- Kuganda kwa chembe nyekundu za damu za mgojwa wa sickle cell kunaweza pia kusababisha kuziba mishipa ya damu hivyo kusababisha Kiharusi.
- Kuganda kwa damu (embolus) kwenye mishipa ya damu ya sehemu nyingine za mwili au (hewa, mafuta, kusanyiko la vijimelea kama bakteria waletao ugonjwa wa endocarditis) huweza
- kusababisha kuziba kwa mishipa inayosambaza damu kwenye ubongo na hivyo kusababisha kiharusi.
- Upungufu wa usafirishaji damu mwilini (systemic hypoperfusion) - Wakati fulani moyo hushindwa kusukuma damu vizuri kwenda sehemu mbalimbali za mwili ikiwemo ubongo kutokana na kufa kwa
- sehemu za nyama ya moyo kwa kukosa damu ya kutosha (Ischaemic Heart Diseases) au kutokana na kujaa kwa maji kwenye gamba lake la nje (pericardial effusion) au kupungua kwa damu mwilini,
- kunakoweza kusababishwa na mambo kadhaa. Hali hii husababisha sehemu kubwa ya mwili ikiwemo ubongo kukosa oksijeni ya kutosha na kusababisha kuathiriwa kwa sehemu ya ubongo.
- Shinikizo la damu la muda mrefu lisilothibitiwa linaweza kusababisha kupasuka kwa mishipa midogo inayosambaza damu kwenye ubongo na hivyo damu kujaa kwenye ubongo (Intracerebral hemorrhage).
Dalili za kiharusi
Dalili za kiharusi hutegemea na eneo la ubongo liliathiriwa.
- Iwapo sehemu ya ubongo mkubwa ( Cerebellum) itaadhirika, mgonjwa atakuwa na dalili kama kushindwa kutembea vizuri, hivyo kuathiri mwendo wake, kujihisi kizunguzungu na pia kutapika.
- Iwapo sehemu ya ubongo wa kati (cerebral cortex) itakuwa imeathirika, mgonjwa atakuwa na dalili za kushindwa kuongea,kushindwa kuelewa lugha inayozungumzwa, kushindwa kuona vizuri, kuwa
- na ukosefu wa kumbukumbu, kuwa na mvurugiko wa mpangilio wakw wa kufikiri, kuchanganyikiwa, na kubadilika kwa mwendo wa harakati za hiari.
- Iwapo mgonjwa ataadhirika sehemu ya ubongo kwenye shingo kuelekea kwenye uti wa mgongo (brain stem), mgonjwa anaweza kuwa na dalili za kuhisi mabadiliko ya harufu, ladha, kusikia na
- kuona; kulegea kwa misuli ya macho (ptosis); kupungua kwa ufahamu na kulegea kwa misuli ya uso; Ulegevu wa Ulimi (kushindwa kutoa nje au kusogeza upande upande); kupungua uwezo wa
- kumeza; ulegevu wa misuli ya shingo na kushindwa kugeuza shingo upande mmoja; kushindwa kusimama sawasawa na kuona vitu kwa hali ya utofauti; mabadiliko ya upumuaji na kiwango cha moyo kudunda.
- Iwapo sehemu mojawapo ya mfumo mkuu wa neva (central nervous system) imeathirika, mgonjwa atakuwa na dalili za kupoteza ufahamu kwa upande mmoja wa mwili na kulegea kwa misuli ya uso, kuhisi ganzi mwilini, na kupungua kwa ufahamu wa hisia na hisia mtetemo.
- Aidha bila kujalisha eneo lililoathiriwa, mgonjwa pia anaweza kuwa na dalili za kupoteza fahamu, maumivu makali ya kichwa na kutapika. Dalili hizi za kuumwa kichwa na kutapika kwa kawaida
- hutokea kwa mgonjwa mwenye kiharusi cha kuvuja damu (hemorrhagic stroke) ambacho husababisha ongezeko la shinikizo na mgandamizo wa ubongo ndani ya fuvu kutokana na kuvuja kwa damu.
Vipimo
- Mionzi
- CT-scan
- MRI
- PET
- SPECT
ECG, ECHOCARDIOGRAM huwezesha kutambua hitilafu katika mapigo ya moyo (arrhythmia) na kama kuna damu iliyoganda kwenye moyo ambayo inaweza kufika kwenye ubongo
Holter monitor husaidia kutambua hitilafu katika mapigo ya moyo (arrhythmia) zinazotokea kwa vipindi
Angiogram huwezesha kugundua matatizo kwenye mishipa ya damu, na ni mishipa ipi ya damu iliyoziba.
Vipimo vya damu huwezesha kutambua uwepo wa lijamu mwilini (hypercholesterolemia) na mabadiliko mengine katika damu
Vihatarishi vya kiharusi
Vitu vinavyoweza kumuweka mtu katika hatari ya kupata kiharusi ni pamoja na
- Shinikizo la damu lisilothibitiwa
- Kisukari
- Uvutaji sigara
- Unywaji pombe kupita kiasi
- Kutofanya mazoezi kabisa
- Fetma (obesity)
- Kuwa na cholesterol nyingi kwenye damu
- Atrial fibrillation
Matibabu ya kiharusi hutegemea pia aina na visababishi vyake .
1. Matibabu ya kiharusi kinachotokana na ubongo kukosa hewa (Ischemic stroke)
Mgonjwa hutibiwa kwa kutumia dawa za kuyeyusha damu iliyoganda (thrombolytics) au kwa kuondoa damu iliyoganda kwa njia mbalimbali (thrombectomy). Dawa nyingine kama vile junior Aspirin na Clopidogrel hutolewa kwa ajili ya kuzuia chembe sahani kukusanyika na kuganda.
2. Matibabu ya kiharusi kinachotokana na damu kuvujia kwenye ubongo (Hemorrhagic stroke)
Aina hii ya kiharusi huitaji tathmini ya upasuaji wa neva ili kuchunguza na kutibu sababu ya damu kuvuja. Angalizo: ni hatari kumpa mgonjwa wa aina hii ya kiharusi dawa za kuyeyusha damu
iliyoganda au za kuzuia kuganda maana uhatarisha maisha ya mgonjwa badal ya kumsaidia. Kwahiyo ni vizuri kwa wataalamu kufanya ufanya uchunguzi wa kutosha ili kuwa na uhakika na tatizo.
Huduma na Matunzo kwa mgonjwa wa Kiharusi
Mojawapo ya mambo muhimu ya kufanya kwa mgonjwa aliyepata kiharusi ni kumuongoza na kumuelimisha ili arudishe ujuzi wake wa maisha ya kila siku. Hapa uhitaji ushirikiano wa wauguzi,
wataalamu wa viungo, wataalamu wa ushauri wa kazi na daktari. Kuna umuhimu pia wa kuwaelimisha ndugu kuhusu hali ya mgonjwa ili waweze kumsaidia katika matunzo yake nyumbani na kwenye jamii inayomzunguka.
Matarajio (prognosis)
Asilimia 75 ya wagonjwa wa kiharusi wanaonusurika kifo huwa walemavu na kusababisha kuathirika kwa ufanyaji kazi wao na kuajiriwa. Ulemavu unaweza kuwa wa kimwili au kiakili au vyote kwa pamoja.
Ulemavu wa kimwili ni pamoja na
- Ulegevu wa misuli
- Kujihisi ganzi sehemu mbalimbali za mwili hususani zilizoathirika
- Kujikojolea
- Kutoona vizuri
- Kushindwa kuendelea na shughuli za kila siku
- Vichomi
- Vidonda shinikizo
- Kukosa hamu ya chakula.
- Ulemavu wa akili hujumuisha vitu kama
- Mgonjwa kuwa na wasiwasi
- Hofu ya mashambulizi, na
- Unyongovu.
Jinsi ya kuzuia kiharusi
- Zuia au tibu shinikizo la damu
- Dhibiti kisukari
- Fanya mazoezi
- Acha kuvuta sigara
- Punguza uzito
- Acha kunywa pombe kupita kiasi
- Tumia Junior Aspirin
- Kula chakula kisichokuwa na mengi na kisicho na chumvi nyingi.
- Tumia dawa za kupunguza mafuta mwilini (statins)kwa mfano Simvastatin
===
What Is a Stroke?
Stroke is a medical emergency and a leading cause of death in the U.S. It occurs when a blood vessel in the brain bursts or, more commonly, when a blockage develops. Without treatment, cells in the brain quickly begin to die. The result can be serious disability or death. If a loved one is having stroke symptoms, seek emergency medical attention without delay.
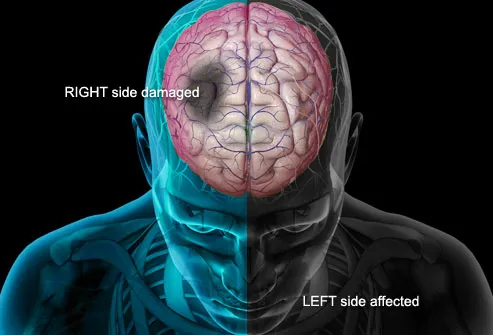
Stroke Symptoms
Signs of a stroke may include:
- Sudden numbness or weakness of the body, especially on one side.
- Sudden vision changes in one or both eyes, or difficulty swallowing.
- Sudden, severe headache with unknown cause.
- Sudden problems with dizziness, walking, or balance.
- Sudden confusion, difficulty speaking or understanding others.
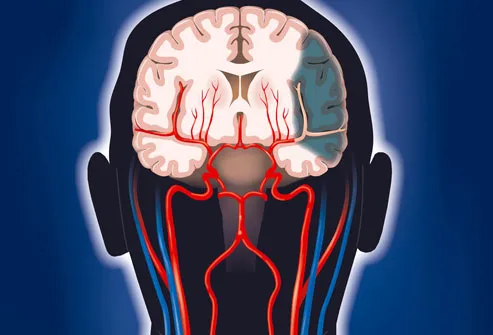
Stroke: Time = Brain Damage
Every second counts when seeking treatment for a stroke. When deprived of oxygen, brain cells begin dying within minutes. There are clot-busting drugs that can curb brain damage, but they have
to be used within three hours of the initial stroke symptoms. Once brain tissue has died, the body parts controlled by that area won't work properly. This is why stroke is a top cause of long-term disability.
